
To stay up-to-date about the latest happenings in the 3D printing world, like us on Facebook or follow us on LinkedIn and Twitter. Visit our Tech News page for more updates on Global 3D Printing News. The portfolio also includes the RESOMER® line of bioresorbable filaments, powders and granules for implantable medical devices, in addition to VESTAKEEP® i4 3DF and VESTAKEEP® Care M40 3DF,Ībout Manufactur3D Magazine: Manufactur3D is an online magazine on 3D Printing. The specialty chemicals company has the industry’s most comprehensive portfolio of 3D printable biomaterials for medical technology, which can be used to make medical device parts that come into contact with the body on a temporary or permanent basis. Since the product launch of the first PEEK filament for 3D printed implants a good three years ago, we have been expanding the possibilities of modern medical technology in the individual treatment of patients using additive manufacturing by constantly developing new innovative biomaterials.” – Marc Knebel, Head of Medical Systems at Evonikįor more than 20 years, Evonik has been the world’s leading manufacturer of high-performance polymers for additive manufacturing technologies. “No other application field showcases more the classic advantages of 3D printing, such as individualization or design freedom, than medical technology. Advancing 3D Printing in Medical Technology  
VESTAKEEP® iC4800, like all Fusion products, is manufactured under strict quality control for biomaterials. Furthermore, the functional additives are available directly on the surface of the 3D printed implant without any additional post-processing steps, which is a first for osteointegrative PEEK biomaterials. Customer feedback and tests on various 3D printers confirm Evonik’s new filament’s excellent processability. For modern medical technology, the development of our first 3D-printable implant. For PEEK materials, they can be printed directly in standard FFF 3D printers. Sold under the brand name VESTAKEEP i4 3DF, this PEEK filament is an. The PEEK filament in natural colour has a diameter of 1.75 mm and is wound onto 250 gramme or 500 gramme spools. Filaments Industrial PVDF (Polyvinylidene Fluoride) Apium PVDF (Polyvinylidene fluoride) Weight: Add to Cart. The VESTAKEEP® iC4800 3DF was created for use in Fused Filament Fabrication (FFF). 3D-Printers & Filament Dryer Industrial Apium P220. This will hasten bone fusion and, as a result, patient recovery. The BCP additive helps bone cells adhere to implants more quickly, which has a positive impact on the bone-implant interface, also known as osteointegration. Biphasic calcium phosphate, a functional special additive, was used to achieve osteoconductivity (BCP). INFINAM PEEK VESTAKEEP i4 3DF VESTAKEEP i4 3DF-T. Excellent biocompatibility and biostability, as well as improved osteoconductive properties, distinguish the new high-performance polymer. In order to better meet the market and the customers needs, IEMAI 3D Printers are compatible with. Advancing 3D Printing in Medical TechnologyĮvonik’s VESTAKEEP® Fusion product line launched in 2020, and the new PEEK filament for 3D printed implants is a biomaterial from that very line.Marc Knebel, head of Evonik’s Medical Devices & Systems market segment, said, “Innovative developments like our VESTAKEEP i4 3DF PEEK filaments are designed for the utmost biocompatibility, biostability and x-ray transparency – making them excellent materials for orthopedic and maxillofacial surgery. It is the world’s first 3D-printable filament to meet this requirement for medical use. Created by medical device company Curiteva, the implant is FDA-cleared and reported to be the world’s first 3D-printed, fully interconnected porous polyether ether ketone (PEEK) implanted structure of its kind for commercial use.ĭesigned especially for use in additive manufacturing processes, Evonik’s VESTAKEEP i4 3DF comes in filament form and meets the requirements of ASTM F2026, which is the standard for PEEK polymers approved for use in surgical implant applications. 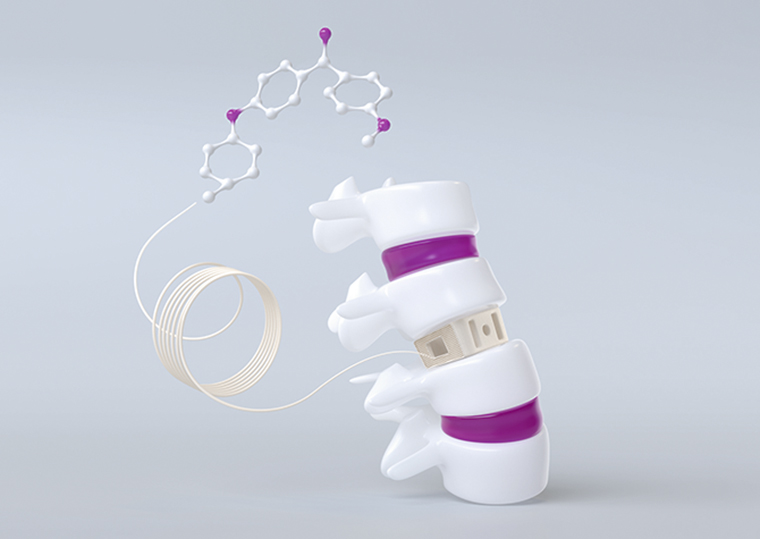
surgeries involving a spinal implant made from Evonik’s VESTAKEEP i4 3DF PEEK filament biomaterial.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
